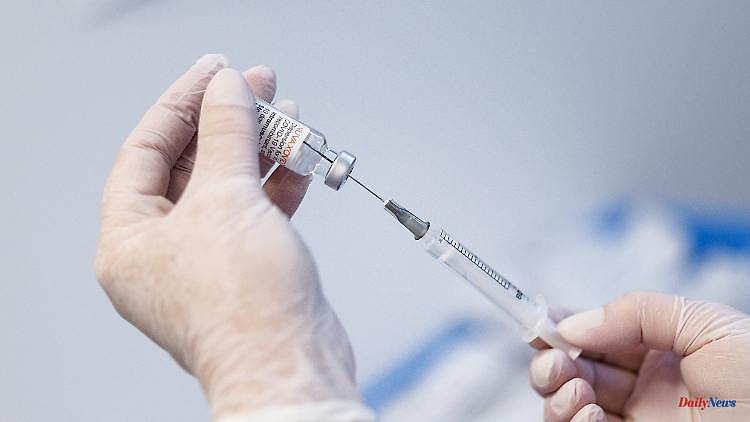
The Novavax vaccination is actually seen as a way of convincing people who are skeptical about vaccination. But that could now change: Cases of heart muscle inflammation have occurred during clinical trials with the vaccine.
The US drug agency FDA is concerned about a possible risk of heart muscle inflammation with the Corona vaccine Novavax. On Friday, the authority published an analysis of the data from the clinical trials with the new vaccine. According to this, six cases of heart muscle inflammation occurred in test persons who received the vaccine. In the group that received only a placebo, there was only one case. A total of 40,000 people took part in the studies.
"The identification of multiple cases that may be associated with the vaccine is of concern," the FDA said. Accordingly, the risk of so-called myocarditis could be higher with Novavax than with the mRNA vaccines from Biontech/Pfizer and Moderna. In the case of the latter, the risk of heart muscle inflammation was found, especially in young men - but only after the clinical trials, as the FDA emphasized.
An FDA independent panel of experts is scheduled to meet on Tuesday and assess the risk of the Novavax vaccine. After the announcement, the manufacturer's shares fell by 20 percent. However, the company stated: "We believe that there is insufficient evidence to establish a causal relationship between the cases of myocarditis and the vaccine."
The Novavax vaccine has already been approved in numerous European countries. The vaccine is protein-based and differs from other vaccines approved in the EU, which are either so-called mRNA or vector vaccines. The authorities originally hoped to be able to convince people who were skeptical about vaccination. However, demand was low.